Corrective Action & Preventive Action Form
The QMSCAPA Corrective Action & Preventive Action (CAPA) Form is organized into eight Tabs or sections. The collection of fields and related Tables are designed to provide tools for the user to record information about an incident or nonconformity, plus information about the cause and corrective actions. 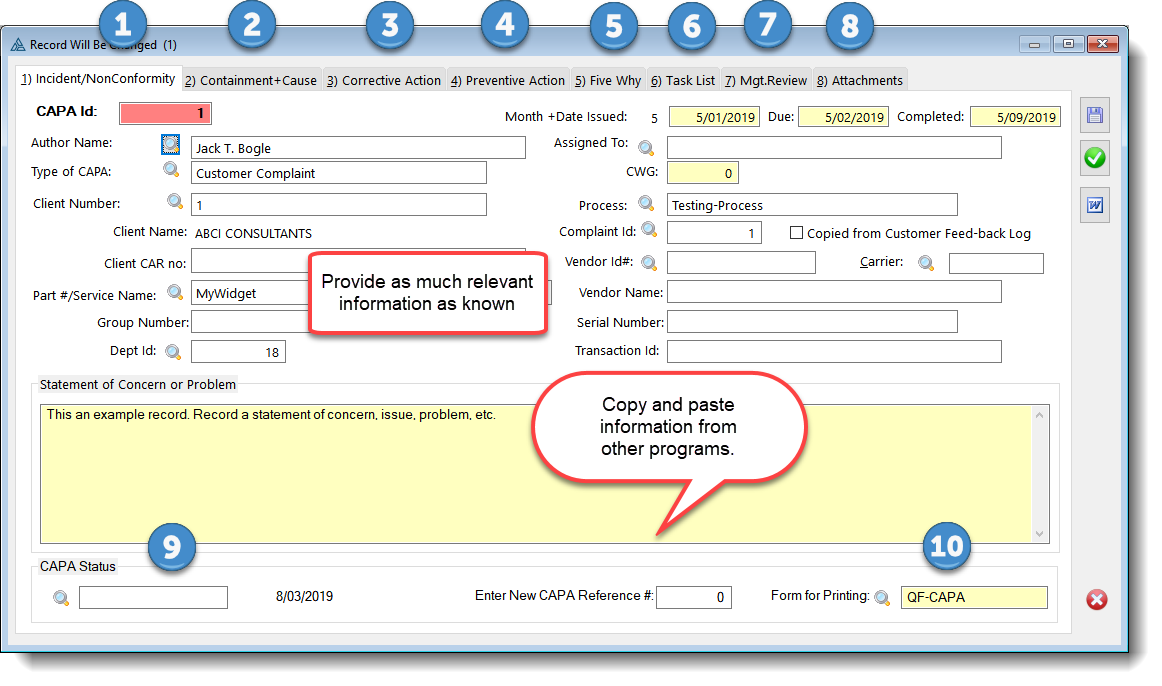 The numeric references in the CAPA form image above are described below:
1)Incident or Nonconformity Statements a.The CAPA Incident or Nonconformity tab is designed to be used for recording information about the incident or nonconformity, including: i.Process / Department ii.Client identifier iii.Part # or Product/Service identifier iv.Group # (reference #, batch #, lot # etc.) v.CAPA Dates (created, saved, issued, due, etc.) vi.Type of CAPA vii.Assignments viii.Origin of the issue, Customer Feed-back (complaint); audit 2)Containment and Cause Statements a.The Containment field is for recording the initial action taken, which is often prior to knowing the full cause or root cause. b.The Statement of Cause field is for recording the information that is often referred to as the 'root cause'. This is a large text field for recording the results or determination of an investigation into the cause of an incident or nonconformity. 3)Corrective Action a.The CAPA Corrective Action tab is designed to be used for recording Corrective Actions. 4)Preventive Action a.The Preventive Action tab is designed to be used for recording Preventive Actions. 5) Five Why a.The Five Why tab contains "5 Why Analysis" for the currently opened CAPA record. 6)Task List a.CAPA Form Task List are designed to organize the investigation and assign others to task that are relevant to the investigation or corrective actions necessary to prevent the incident or nonconformity from being repeated. 7)Management Review a.Management review tab contains comments and review dates, including: b.Verification, proof of effectiveness and the date verified' 8)Attachments a.The CAPA Form Attachments tab is designed to assist with the organization and retrieval of attached (or stored) electronic documents that are related to a CAPA.
|
 +1 800 644 2056
|
